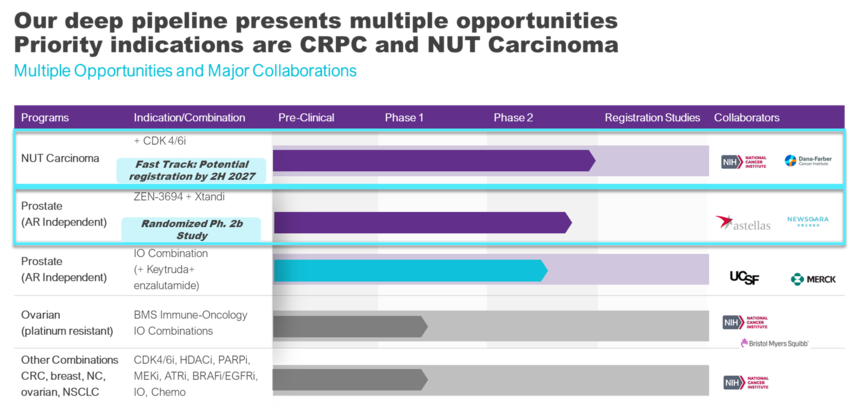
Our development strategy is focused on targeting patient subsets with high unmet need and those that are most likely to benefit from therapy. We are advancing multiple clinical programs using our lead candidate, ZEN-3694, in combination with other drugs in oncology indications. Our two priority indications are:
- ZEN-3694 in combination with a CDK4/6 inhibitor (abemaciclib) for the treatment of metastatic or unresectable NUT carcinoma in patients who have received at least one line of prior chemotherapy. This program received FDA Fast Track designation.
- ZEN-3694 in combination with Astellas and Pfizer’s enzalutamide for the treatment of metastatic castration resistant prostate cancer (mCRPC) in patients that have progressed on an androgen receptor pathway inhibitor (ARPI).
Phase 1 and 2 clinical trials combining ZEN-3694 with immune checkpoint inhibitors, HDAC inhibitors, kinase inhibitors, PARP inhibitors, and chemotherapy in various solid tumor indications are also in progress.